Laboratory sanitization in Seattle isn’t optional-it’s a legal requirement. Federal regulations from the EPA and OSHA set strict standards, and Washington State adds its own rules on top of that.
We at Bumble Bee Cleaning Services work with labs every day, and we’ve seen firsthand how compliance mistakes lead to costly penalties. This guide walks you through what you need to know and how to get it right.
Understanding Laboratory Sanitization Standards in Seattle
Federal Regulations Set the Foundation
Federal law establishes baseline requirements for laboratory safety, but Seattle labs operate within a layered regulatory environment that extends far beyond what OSHA and the EPA mandate. OSHA’s laboratory standard, 29 CFR 1910.1450, requires a written Chemical Hygiene Plan, personal protective equipment, and hazard communication for chemical exposures. The EPA regulates waste disposal under RCRA and hazardous waste classifications, meaning your lab’s chemical and biological waste streams must follow specific protocols. Washington State enforces these standards through the Department of Health, which conducts unannounced inspections and imposes penalties ranging from $7,000 to $70,000 per violation for non-compliance.

Cannabis Testing Labs Face Additional Requirements
If your lab handles cannabis testing, Washington’s cannabis testing program under WAC 314-55-090 requires a complete Standard Operating Procedure manual that details decontamination, cleaning procedures for instruments and bench space, and cannabis waste disposal following WAC 314-55-097. Your scientific director must review and authorize this SOP, with all changes documented and available to staff in their work areas. A single deviation from your documented procedures without approval in your deviation log can trigger a violation notice. Any lab handling bloodborne pathogens must comply with OSHA’s bloodborne pathogen standard, which requires initial 40 hours of hazardous materials training with annual recertification.
Permits and Inspections Add Operational Complexity
Seattle’s Department of Construction and Inspections requires permits for lab sanitization system upgrades, including Boiler & Pressure Vessel, Electrical, Mechanical, and Building inspections before you can operate new equipment. Non-compliance results in operational shutdowns that cost far more than proper preventive compliance.
Documentation Requirements Exceed Most Labs’ Expectations
Most labs underestimate the documentation required to pass a state audit. Your SOP manual must include not just what you do, but why you do it, with sections covering safety precautions, PPE policies, chemical spill response, access control, sample handling, and employee medical attention procedures. Washington DOH requires that you maintain retired procedures for five years beyond retirement and reconstruct prior procedures on demand. ATP testing for sanitization validation should target readings below 10 relative light units for sterile environments, and you must keep detailed records of these tests. If your lab uses EPA-approved disinfectants like quaternary ammonium compounds or hydrogen peroxide vapor, documentation must show which disinfectant was used, when, and on which surfaces. Deviation logs must record every instance you deviated from your procedures, the reason, and the scientific director’s approval-missing this single document can result in a violation. Your lab waste disposal documentation is equally important: you must maintain chain-of-custody procedures, waste-stream segregation records, and evidence that disposal was handled by a licensed facility. These records must be available within minutes during an inspection, not buried in filing cabinets.
Gaps in Documentation Create Operational Risk
Labs that discover documentation gaps only during an inspection face weeks of operational delays while they reconstruct records. The inspection process moves quickly, and inspectors expect immediate access to complete records. This is where specialized cleaning protocols become valuable-they help labs maintain the records that prevent violations. Your next step involves identifying which specific cleaning protocols and documentation systems your lab currently lacks.
How to Build a Cleaning and Sanitization Protocol That Passes Inspection
Document Every Surface You Clean
Your cleaning protocol serves as proof that your lab meets Washington State standards. The difference between passing and failing an inspection often comes down to whether your team can demonstrate what they cleaned, when they cleaned it, and what method they used. High-touch surfaces in your lab-door handles, faucet controls, and instrument keypads-require EPA-approved disinfectants with documented contact times. Quaternary ammonium compounds work well for routine surfaces and typically need 10 minutes of contact time, while hydrogen peroxide vapor handles specialized incidents involving bloodborne pathogens or fentanyl residues more effectively. The CDC supports both approaches, but your choice matters because your SOP must specify which disinfectant you use, on which surfaces, and why.
Your documentation must include the date, time, disinfectant used, contact time, and ATP reading results. Without this record, you have no proof that sanitization occurred, and an inspector will flag it as a violation.
Validate Sanitization With ATP Testing
ATP testing validates that your sanitization actually worked. Try for readings below 10 relative light units for sterile environments, and schedule these tests after every cleaning cycle for high-risk areas. Your team records the date, time, disinfectant used, contact time, and ATP reading results for each test. This documentation becomes your evidence during inspections-inspectors expect to see these records immediately and in organized form.
Establish Instrument-Specific Decontamination Steps
Equipment and instruments demand a different approach than surfaces. Biosafety cabinets and microbial hoods need HEPA filtration systems that capture particles down to 0.3 microns, with filter replacements scheduled every 6 to 12 months depending on contamination levels. Your SOP must detail the specific decontamination steps for each instrument type, including bench space ventilation requirements and the sequence of cleaning steps that prevents cross-contamination between samples.
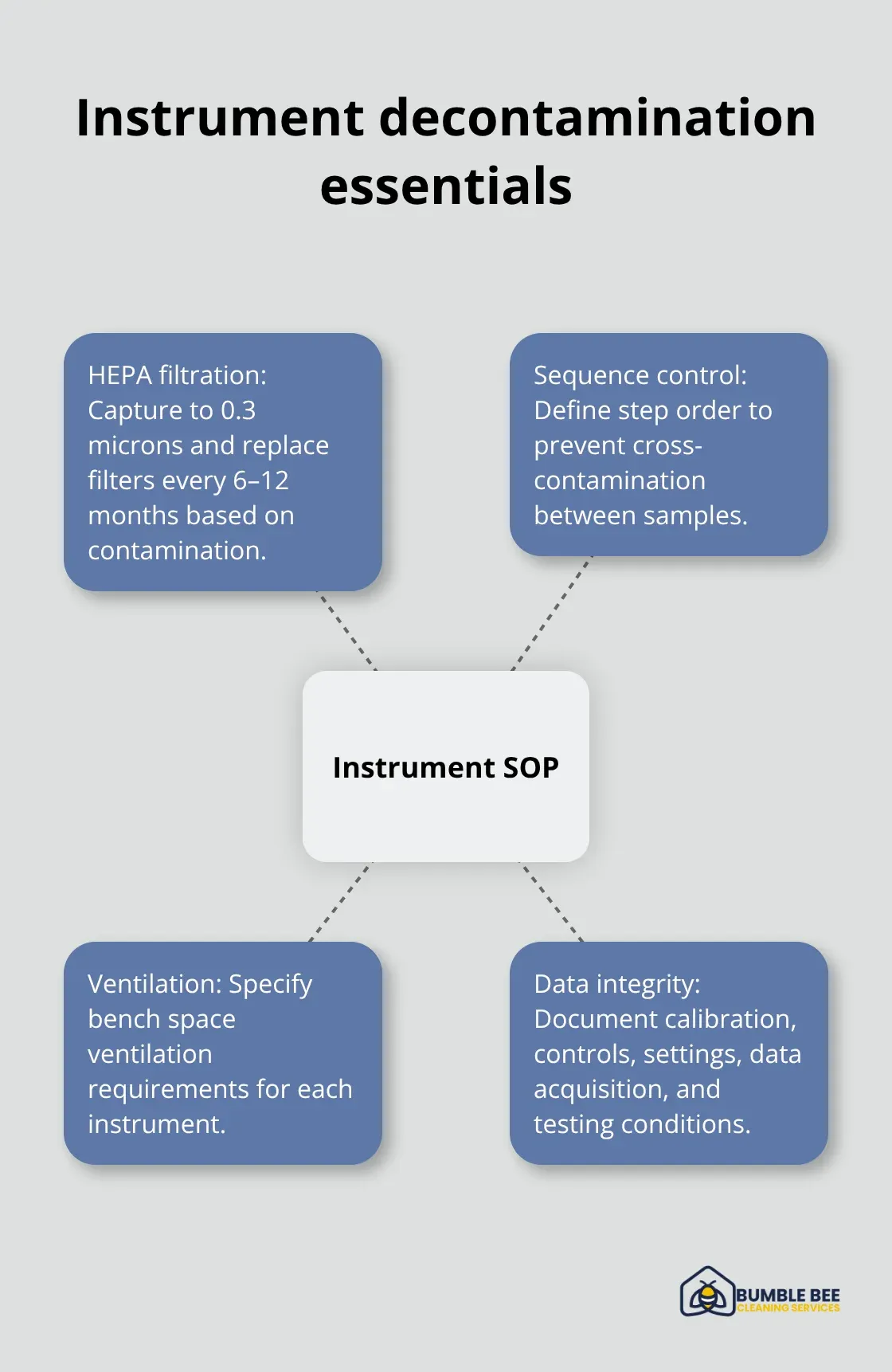
OSHA laboratory safety guidance requires that your team document calibration and control preparation, instrument settings, data acquisition, and testing conditions to maintain data integrity.
Many labs skip this step because it feels administrative, but inspectors expect to see these records immediately. Your deviation log is equally important-it must record every instance where your team deviated from documented procedures, the reason for the deviation, and the scientific director’s approval. A single missing entry in this log can trigger a violation notice.
Maintain Chain-of-Custody Records
Chain-of-custody procedures for sample and reagent usage, storage conditions, and waste disposal must include timestamps and staff initials. The goal is reconstruction: if an inspector asks what happened to a specific sample on a specific date, your documentation should answer that question within seconds. These records demonstrate that your lab maintains control over samples from collection through disposal, which satisfies both state auditors and federal oversight requirements.
Your next step involves identifying which specific documentation systems your lab currently lacks and establishing the protocols that prevent violations before inspectors arrive.
Who Should Handle Your Lab Sanitization
General Cleaning Companies Fall Short
When your lab faces a state inspection, the cleaning service you select determines whether you pass or fail. This isn’t about finding the cheapest option-it’s about finding a partner who understands Washington’s regulatory framework and documents every step they take. Most general cleaning companies lack the specialized knowledge required for laboratory environments. They don’t understand EPA-approved disinfectants, they’ve never validated sanitization with ATP testing, and they can’t help you maintain the chain-of-custody records that inspectors demand.
Certifications and Hands-On Experience Matter
You need a cleaning service with IICRC biohazard remediation training, hazmat operations certification, and demonstrated experience with bloodborne pathogen protocols. The Washington State Department of Health mandates that staff complete 40 hours of initial hazardous materials training, followed by annual recertification for anyone handling high-risk lab environments. When you request certifications, verify that trainers completed their coursework within the last two years-outdated credentials suggest the company isn’t staying current with regulatory changes.
Call labs that have passed recent state audits and ask specifically whether the cleaning service helped them maintain documentation systems and prepare for inspections. A service that simply cleans surfaces without helping you build sustainable documentation practices will leave you vulnerable during audits.
Equipment and Real-Time Validation
Your cleaning partner should provide real-time ATP testing results with acceptable readings below 10 relative light units for sterile environments, and these results should integrate automatically into your records system. Electrostatic sprayers and negative air machines represent the technical baseline for professional lab work-equipment costs typically run $3,000 to $5,000 for electrostatic units and $2,000 to $4,000 for negative air machines. Verify that your service provider owns this equipment rather than renting it sporadically.

Proactive Compliance Planning
Request a detailed SOP review from your cleaning service before they begin work. They should identify gaps in your current documentation, propose specific protocols for high-touch surfaces and instrument decontamination, and establish a schedule for ongoing ATP validation testing. The service should also conduct quarterly audits of your lab’s compliance readiness and provide written reports that identify potential violations before inspectors arrive.
This proactive approach costs more upfront but prevents the $7,000 to $70,000 per violation penalties that reactive cleaning creates. Don’t accept vague promises about compliance-ask for the specific documentation systems they’ll implement, the frequency of ATP testing, and how they’ll integrate their work with your deviation logs and chain-of-custody records. A cleaning service that can’t articulate these details won’t protect your lab when inspectors arrive.
Final Thoughts
Laboratory sanitization in Seattle demands more than surface-level cleaning-it requires sustainable protocols, ATP validation, and organized records that satisfy state auditors before inspectors arrive. The labs that pass inspections build proactive compliance systems rather than react after violations appear, and they partner with cleaning services that understand Washington’s regulatory framework and document every step. When you select a cleaning partner, verify their IICRC biohazard remediation training, hazmat operations certification, and hands-on experience with bloodborne pathogen protocols, then ask them to review your SOP for gaps and establish quarterly compliance audits.
Your next step involves scheduling a consultation with a cleaning service that specializes in laboratory environments and can explain exactly how they’ll integrate ATP testing, equipment decontamination, and chain-of-custody documentation into your lab’s operations. We at Bumble Bee Cleaning Services work with Seattle labs to maintain compliance and prevent the $7,000 to $70,000 per violation penalties that reactive approaches create. Visit our website to discuss your lab’s specific laboratory sanitization needs and build a proactive compliance plan that protects your operation from violations and operational shutdowns.
